We have a new paper just published in the Journal of Neurophysiology! In it, we explored the phenomenon of saccadic inhibition from a multisensory perspective. Specifically, when a sudden visual stimulus appears in the environment, saccade generation rhythms are reflexively interrupted, and with a very short latency. One of our long term goals is to understand the underlying mechanisms for such interruption.
In this new study, we paired visual stimuli with spatially-uninformative sounds. The idea was to use the sounds to alter temporal processes associated with saccadic inhibition, but not influence spatial representations (like where the sensory stimuli are in the environment) in a strong way. This goes back to our hypothesis that there might be independent spatial and temporal components to saccadic inhibition mechanisms (like here, here, and here).
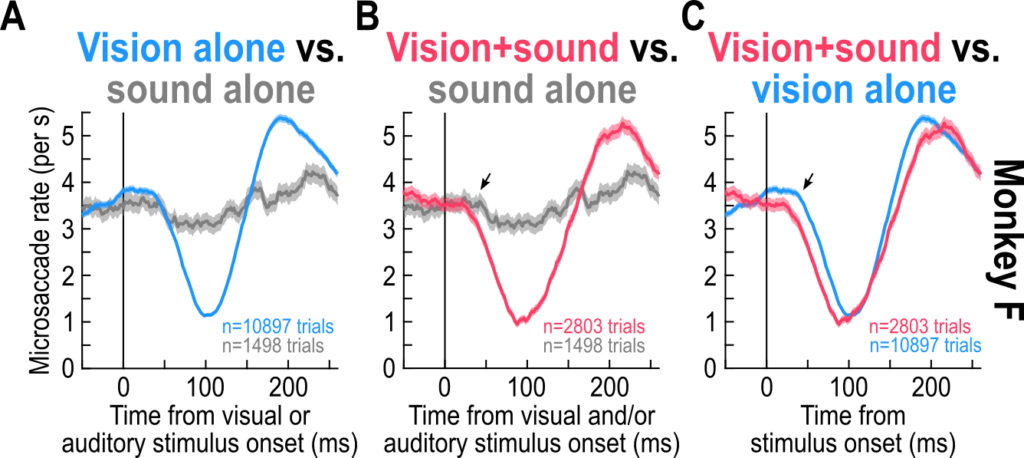
We found that sounds alone had a minimal influence on saccade generation rhythms. Nonetheless, when paired with visual stimuli, they still accelerated the visually-driven inhibition. This suggests a multisensory integration process that is beyond superposition, and it might indicate that the oculomotor system can access multisensory stimuli with a privileged way. This is important for our follow-up neurophysiological investigations of the sensory-motor pathways driving saccadic inhibition.
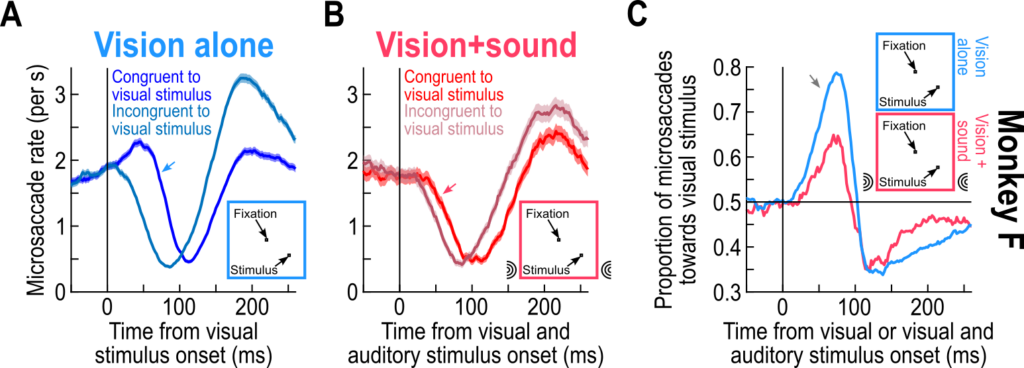
Importantly, we had known for many years that eye movement directions are also altered by visual stimuli (like here and here). When we added the sounds in our experiments, whether these sounds were spatially-uninformative or spatially-lateralized, the visually-driven modulations in eye movement directions persisted. Thus, the visual modality dominates the eye movement directions, but the sound can accelerate temporal aspects of saccadic modulations.
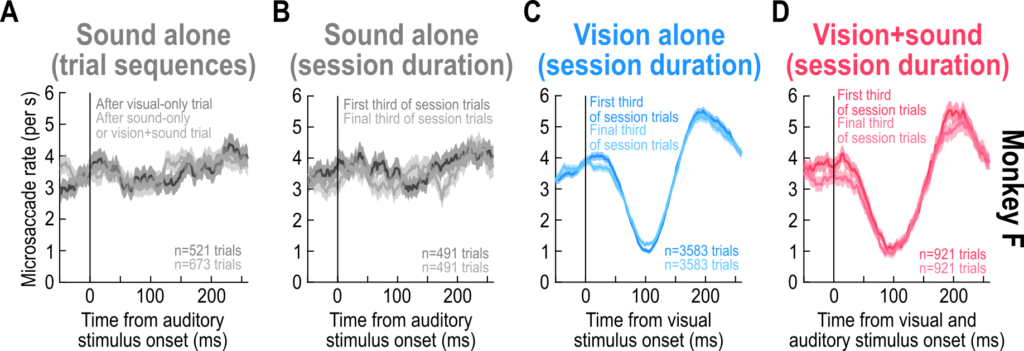
Finally, our experiments allowed us to investigate a fundamental property of saccadic inhibition: whether it adapts away to repeated exposure to the same sensory stimuli. As is known, sensory responses in the brain always tend to adapt to repeated stimulus exposure from the environment. Remarkably, there was no evidence of adaptation in this particular eye movement phenomenon. Thus, with thousands of trial repetitions, saccadic inhibition behaved similarly across trials. This is similar to our earlier casual observations in which we repeated stimuli for up to 10,000 trials, but we could quantitatively document it in the current paper. Moreover, there was no adaptation to the sounds. If anything, there might have been some sensitization to the sounds in some cases.
On the one hand, the lack of adaptation of saccadic inhibition is an experimental blessing. It means that we can pool saccadic inhibition trials from many trial repetitions and sessions when characterizing the phenomenon and its properties. On the other hand, it suggests that the sensory neural responses driving saccadic inhibition either do not adapt like might be expected from classic visually-sensitive circuits (like the visual cortex or superior colliculus), or that the readout of these sensory neural responses gets progressively amplified to allow the behavioral phenomenon of the eye movements to not adapt. This constrains the brain regions that might be implicated in driving saccadic inhibition, and we continue to investigate these brain regions.
In our follow up work, we will use the paradigms described in the current study to explore the different sensory and motor pathways that might be implicated in the reflexive phenomenon of saccadic inhibition. This can have broader implications than just about this oculomotor reflex, because it can allow us to delineate the differential roles of anatomically parallel routes for sensory signals to ultimately reach the brain’s cognitive and motor systems – a fundamental problem in systems neuroscience for many decades.
