
Our lab participated in this year’s ZüTüMü conference, which took place in Bern, Switzerland. The conference covered a wide range of topics related to perception, oculomotor control, and vestibular systems.
A hallmark of the conference is that it brings together expertise from clinician scientists as well as fundamental scientists. This gives us a flavor of the implications of our scientific discoveries for human health.
Our lab participated with three posters and two talks at this conference:
1) The neural basis of pupil diameter dynamics
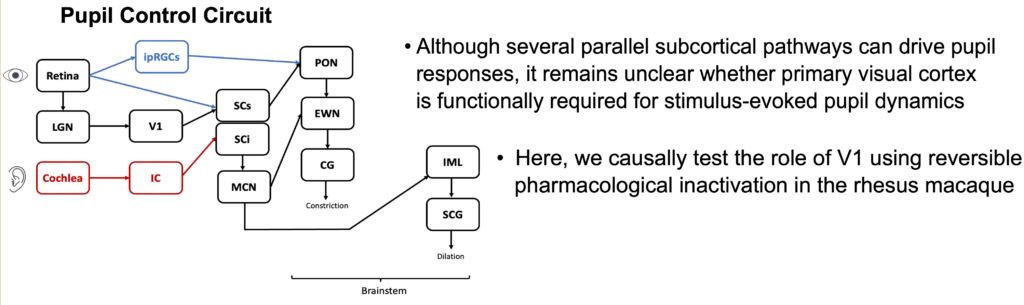
In the first poster, Ekaterina described very exciting results in which we inactivated a portion of the primary visual cortex (V1) and investigated how stimulus-evoked pupil diameter dynamics are affected. Importantly, we tried both auditory stimuli and visual stimuli (as well as multisensory audio-visual stimulation). Remarkably, even pupil responses to auditory stimuli, in the complete absence of visual stimuli, were affected by V1 inactivation!
2) Fine control of rapid eye movements by omnipause neurons
In the second poster, Maria looked at how brainstem omnipause neurons (OPN’s) gate saccadic eye movements. It has been known for decades that these neurons “pause” their activity during saccades of all sizes and directions. However, Maria looked at the smallest possible saccades (including microsaccades). Remarkably, we found that the pause in OPN’s is not as complete for tiny saccades as it is for larger eye movements. This is truly remarkable because it shows fine-grained control by OPN’s on different eye movement types. Indeed, these results are, in our view, the very first physiological correlate of known excitatory projections from the foveal quarter of the superior colliculus (SC) to OPN’s. For many years, these excitatory projections were thought to prevent OPN’s from pausing. However, this has created a conundrum because the OPN’s clearly pause for small saccades (or at least, reduce their tonic firing rate). Our results suggest that the OPN pause is modulated for small saccades, such that it is not complete, and that the excitatory projections from the small-saccade portion of the SC to the OPN’s are not necessarily there to prevent saccades from happening – that portion of the SC actually drives small saccades. Rather, the excitatory projections are revealed via an increased likelihood of intra-saccadic spiking by OPN’s for small saccades. We hope to develop models of what this means for the control of saccadic eye movements by brainstem circuitry.
3) Prediction of saccadic eye movement timing variability by SC visual responses, but not by V1 visual responses
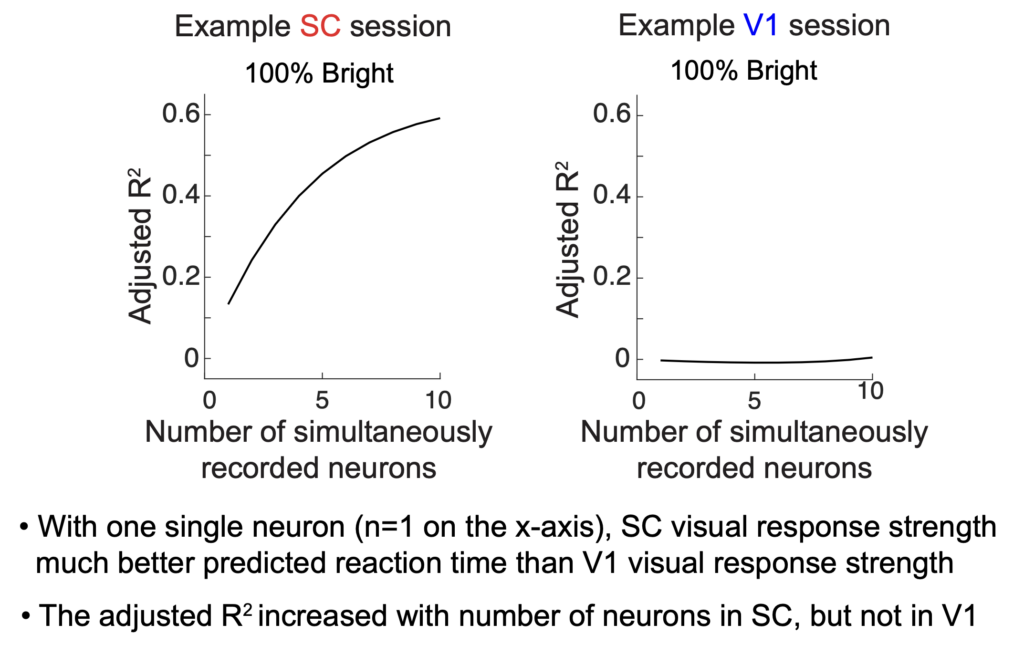
The third poster was presented by Yue, and it was a follow-up to a recent paper of ours. Specifically, the SC receives a large portion of its visual input from V1. However, does this mean that SC visual responses serve the same function as V1 visual responses? We think not. We analyzed the trial-by-trial variability of SC visual response strength, and we correlated it with trial-by-trial variability of saccadic eye movement reaction time. We knew from our previous work that for any individual neuron, it was more likely for the SC visual response strength to exhibit a correlation with saccadic reaction time (stronger visual burst implies shorter saccadic reaction time), and we also knew that this was not the case for V1 visual responses. Here, we exploited the fact that we could record from simultaneous SC neurons during the same behavior, as well as simultaneous V1 neurons in other sessions. So, we made very simple linear models for predicting trial-by-trial saccadic reaction time variability based on a linear combination of the visual response strengths of N simultaneously recorded neurons (in either the SC or V1). We then checked the prediction quality of the saccadic reaction time as a function of how many simultaneous neurons were included in the prediction. As expected, the prediction quality increased with the number of simultaneous neurons in the SC. Remarkably, this was generally not the case in V1!!! Moreover, with only one single neuron in the prediction, the SC prediction was much higher than in V1 as we showed in our earlier work. Thus, SC visual responses are a transformed version of the visual inputs to the SC, and in such a way that aids in jumpstarting the orienting response mediated by the SC.
4) Selective saccadic suppression of bright visual information in V1, but not SC, neurons
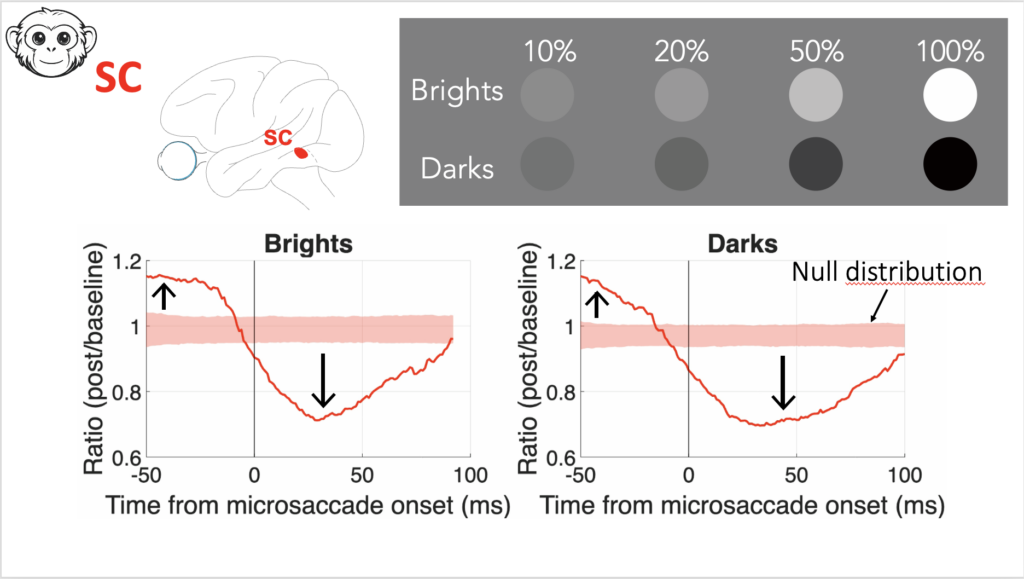
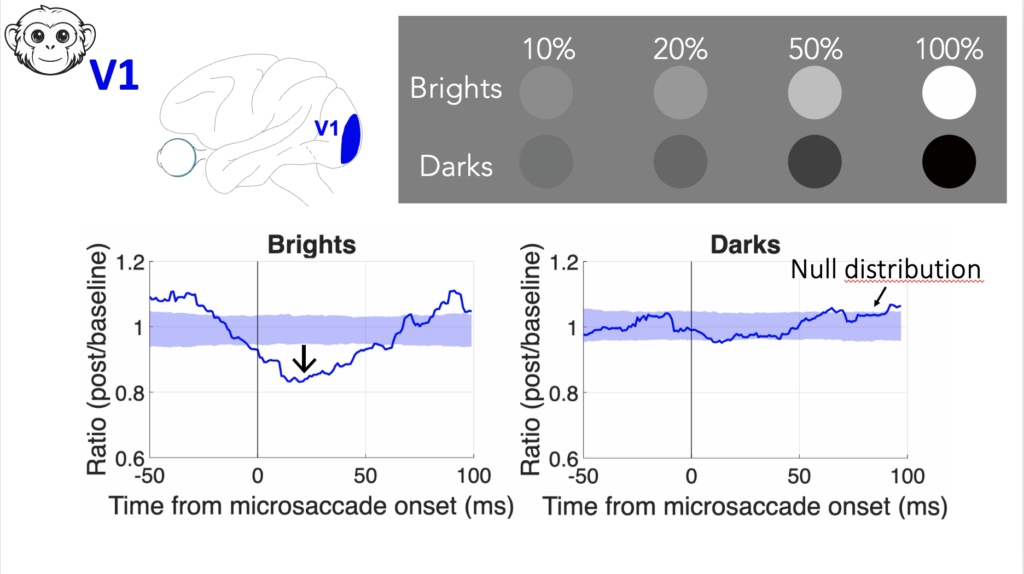
For the talks, one was presented by Wenbin. He looked at the phenomenon of reduced visual sensitivity if stimuli come around the time of saccadic eye movements. He specifically looked at how such “saccadic suppression” depends on the visual appearance of the visual images. We had known from our prior work that in perception, saccadic suppression causes the same perceptual deficit if a target was bright or dark. The question, then, was whether this reflects neural modulations in V1 or in the SC. Remarkably, we found that perception reflects the neural modulations of the SC, and not V1! This suggests a deviation of V1 representations from perception. Wenbin then described how changing the task requirement to one involving discrimination can still favor readout of V1 activity. This reveals a remarkable division of labor between different brain areas for supporting behavior.
5) Revisiting the action potential waveform from the perspective of the SC
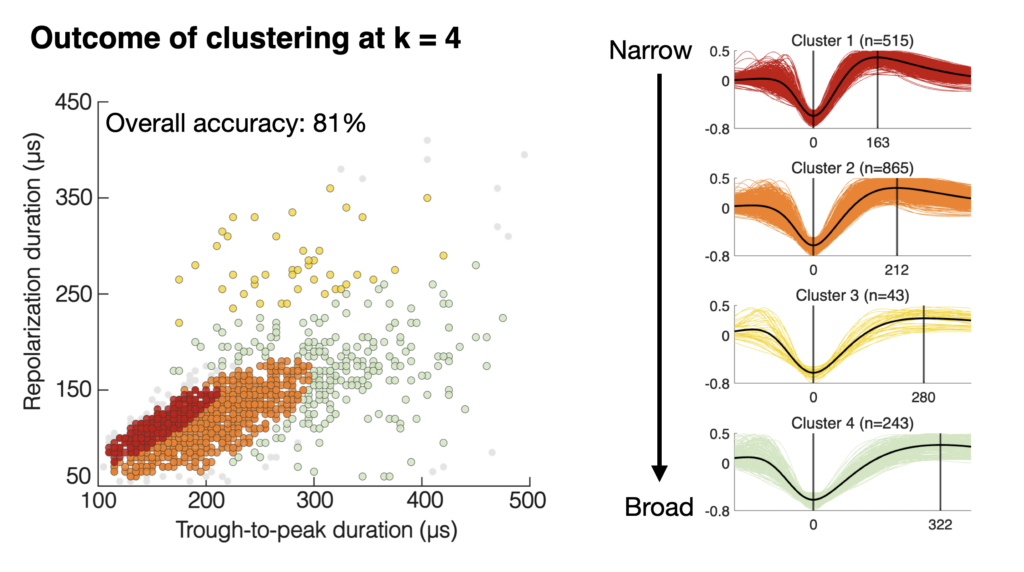
Finally, Shweta gave a talk about action potential waveforms. There have been discussions in the literature about how many classes of action potential waveforms may exist (based on a pair of width parameters related to membrane depolarization and subsequent repolarization). However, these works were all primarily cortex-centric. In our work, Shweta looked at the waveforms of thousands of SC neurons, and she compared them to those of similar numbers of V1 neurons. The advantage of the SC is that we have well-known functional classifications of its cell types. Shweta found that not all waveform classifications are the same across brain areas, with the SC showing much narrower waveforms (in general) than in the cortex. Even in V1 itself, the classifications are different from those in higher-level cortical brain areas. These results are a useful way for relating cell types to extracellular measurements, and they also highlight how other factors, like recording technology, will matter a great deal for interpreting action potential waveform shape.






